How structural biology is reshaping tuberculosis research and treatment
March 24, 2026
Tuberculosis is an ancient disease that still impacts human health today.
Tuberculosis is a disease of the lungs that also can affect many other parts of the body including the kidneys, spine and brain. Symptoms include severe cough, extreme chest pain, weight loss, fever, chills, loss of appetite and night sweats. Without treatment, tuberculosis can be deadly.1
For much of history, the source of tuberculosis was unknown. That changed in 1882 when Dr. Robert Koch identified Mycobacterium tuberculosis as the bacterium that causes the disease.2
Decades of research has resulted in effective treatments that save lives. Despite these critical advances, tuberculosis remains among the top 10 causes of death, globally. One reason is that the bacteria has evolved new ways to survive by growing resistant to current medications. At the same time, existing treatments are long and demanding, requiring people with tuberculosis to take multiple medications every day for six to nine months. For many people, even accessing these medications can be incredibly difficult.
One way to address these challenges is to develop new medications that bypass the bacteria’s defenses. One promising approach may be found in a field called structural biology.
“Structural biology helps us visualize the molecules that make us who we are,” said Dr. Huilin Li, the Ralph and Grace Hauenstein Endowed Chair in Structural Biology and Chair of VAI’s Department of Structural Biology. “By understanding how their shape influences their function, we can more precisely pinpoint vulnerabilities in tuberculosis bacteria and develop treatments that target the machinery essential for their survival.”
Li is an internationally recognized structural biologist whose work has revealed vulnerabilities in Mycobacterium tuberculosis that can help combat medication resistance. Previous work by Li has uncovered how the bacterium uses a system called Pup-proteasome to fend off attacks by the body’s immune system.
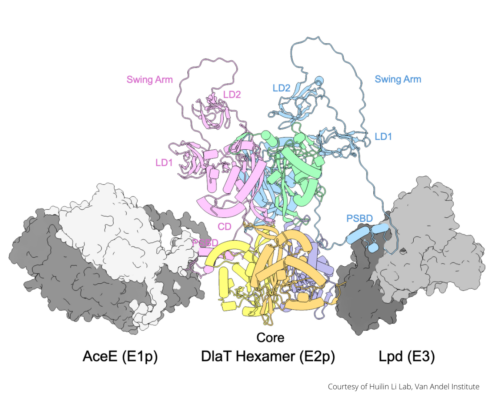
Now, recent findings published in the Journal of Biological Chemistry by Li and collaborators reveal a new vulnerability in Mycobacterium tuberculosis. They found that the structure of an energy processing enzyme complex in the bacterium is built differently than expected. This is important, Li said, because its shape directly impacts how it does its job.
Called pyruvate dehydrogenase (PDH) complex, this enzyme helps cells generate energy. PDH is highly conserved across organisms, meaning it remains nearly identical in most species. Because of this, scientists have long believed PDH to function the same in tuberculosis bacteria as it does in other organisms.
Typically, PDH forms a large soccer ball-shaped structure but, in tuberculosis bacteria, Li and his collaborators found that the PDH complex is broken up into a smaller six-part structure.
“This altered structure appears to be an adaptation that helps the bacterium survive inside the human body,” said Li. “Because tuberculosis has been around for millions of years, it has developed strategies to survive stressful situations and evade existing medications.”
Inside the body, tuberculosis bacteria take refuge within immune cells, which are responsible for neutralizing threats. This ability to hide inside the very cells designed to fight bacteria help Mycobacterium tuberculosis evade immune attack and make it harder for medications to treat them.
Learn more about structural biology research at VAI ➔
The immune cells don’t make it easy. They release chemicals that cause a highly stressful environment for tuberculosis bacteria. However, as discovered by Li and his team, the altered structure of the bacteria’s PDH complex allows it to withstand harsher environments that many other microbes could not.
This structural difference in PDH is a potential weak spot in tuberculosis bacteria and could help explain why the disease is so resilient. However, future research is needed to better understand the architecture of this complex and how it supports the bacteria’s survival.
“In a disease where treatment is lengthy, demanding and challenged by drug resistance, going back to the basic structure of these molecules can be a gamechanger, “Li said. “Structural biology is helping us move toward precision strategies that could lead to more efficient and effective treatments for tuberculosis.”
Funding Acknowledgements
Research reported in this publication was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under award no. R01AI175342 (Bryk) and Van Andel Institute (Li). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Sources
1 Centers for Disease Control and Prevention. (2025 Jan. 17). About Tuberculosis. https://www.cdc.gov/tb/about/index.html
2 Centers for Disease Control and Prevention. (2025 Dec. 5). History of World TB Day. https://www.cdc.gov/world-tb-day/history/index.html

