Cryo-EM reveals ignition mechanism for DNA replication
October 22, 2017

GRAND RAPIDS, Mich. (Oct. 23, 2017) — An international team of scientists, led by scientists at Van Andel Research Institute, Cold Spring Harbor Laboratory and Imperial College London, has shed new light on a critical step in DNA replication, offering fresh insights into a fundamental process of life and driver of many different diseases, including cancer.
“Previous studies have described how enzymes assemble and gather around DNA to prepare it for replication. Here, we describe what these enzymes do to DNA once they are in place,” says Huilin Li, Ph.D., a professor in the Center for Epigenetics and one of the senior authors on a study scheduled for publication this week in Proceedings of the National Academy of Sciences.
The work is part of a long-time collaboration between Li, Bruce Stillman, Ph.D., president of Cold Spring Harbor Laboratory, and Christian Speck, Ph.D., professor at Imperial College London.
DNA replication is a tightly choreographed process that copies the genetic code, allowing its instructions to be passed on from one generation of cells to the next. In diseases like cancer, these mechanisms can fail, leading to uncontrolled or faulty replication with devastating consequences.
“How this complex process starts is not well understood at the molecular level,” Li adds. “Our hope is that the more mechanistic detail we learn about how replication works, the better able others will be in developing new treatments for cancer and other diseases.”
Their discovery wouldn’t have been possible without a revolutionary technology called cryo-electron microscopy, or cryo-EM, which allows scientists to see critical biological components in exquisite, atomic detail. Using the powerful microscopes in VARI’s David Van Andel Advanced Cryo-Electron Microscopy Suite, Li and his collaborators revealed the very first steps in DNA replication—the separation of the two DNA strands from one another.
Before replication can take place, a pair of structures called heterohexameric minichromosome maintenance (Mcm2-7) helicases are assembled head to head on the DNA double helix as a double hexamer. They eventually separate into two functional helicases and, in the process, each push out one strand of the double helix. Later, when DNA replication starts, the two helicases each move on one strand of DNA in opposite directions to unwind the helix.
Computational rendering of the cryo-EM images revealed the three-dimensional structure of these helicase enzymes. Imaging from the team’s study shows the helicase enzymes binding to 60 base pairs of the DNA double helix.
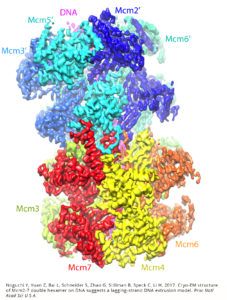
Li likens it to a spring-loaded mechanism that puts pressure on either side of DNA, bending the helix into a zig-zag shape. This positions the DNA strands toward two side-way gates, ready to be pushed out in the next stage when the two Mcm2-7 hexamers disjoin, going opposite directions to “unzip” the double helix.
“These are processes at the very foundation of life that have largely remained a mystery to biologists since the discovery of DNA double helix more than 60 years ago,” says Li. “Thanks to technologies like cryo-EM, we are able to ‘see’ the operational mechanism in action, which gives us valuable knowledge to improve health for people around the world.”
The work is part of a long-time collaboration between Li, Bruce Stillman, Ph.D., president of Cold Spring Harbor Laboratory, and Christian Speck, Ph.D., professor at Imperial College London. Previous studies from the group have evaluated how the helicase first assembles around DNA to prepare for the step described in today’s paper.
For more about how the helicase assembles around DNA, please see ORC as Loader of the Rings: Study details ringed structure of ORC in DNA replication, a February study published in Nature Structural and Molecular Biology.
Authors of today’s paper are: Yasunori Noguchi, Sarah Schneider and Christian Speck, all of Imperial College of London; Zuanning Yuan, Lin Bai, Gongpu Zhao and Huilin Li, all of Van Andel Research Institute; and Bruce Stillman of Cold Spring Harbor Laboratory.
Research reported in this publication was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award number R01GM111742. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
###
ABOUT VAN ANDEL RESEARCH INSTITUTE
Van Andel Institute (VAI) is an independent nonprofit biomedical research and science education organization committed to improving the health and enhancing the lives of current and future generations. Established by Jay and Betty Van Andel in 1996 in Grand Rapids, Michigan, VAI has grown into a premier research and educational institution that supports the work of more than 360 scientists, educators and staff. Van Andel Research Institute (VARI), VAI’s research division, is dedicated to determining the epigenetic, genetic, molecular and cellular origins of cancer, Parkinson’s and other diseases and translating those findings into effective therapies. The Institute’s scientists work in onsite laboratories and participate in collaborative partnerships that span the globe. Learn more about Van Andel Institute or donate by visiting www.vai.org. 100% To Research, Discovery & Hope®
ABOUT IMPERIAL COLLEGE LONDON
Imperial College London, founded in 1907, is the only UK university to focus entirely on science, engineering, medicine and business. Our international reputation for excellence in teaching and research sees us consistently rated in the top 10 universities worldwide. Generations of Imperial staff, alumni and students have changed the world with cutting-edge innovations; from the discovery of penicillin to the world’s first invisibility cloak. Recognition of this ground-breaking work has come in the form of 14 Nobel Prizes, 81 Fellowships from the Academy of Medical Sciences, 77 from the Royal Academy of Engineering and 73 from the Royal Society. For more information, visit www.imperial.ac.uk.
ABOUT COLD SPRING HARBOR LABORATORY
Founded in 1890, Cold Spring Harbor Laboratory has shaped contemporary biomedical research and education with programs in cancer, neuroscience, plant biology and quantitative biology. Home to eight Nobel Prize winners, the private, not-for-profit Laboratory is led by Dr. Stillman and employs 1,100 people including 600 scientists, students and technicians. The Meetings & Courses Program hosts more than 12,000 scientists from around the world each year on its campuses in Long Island and in Suzhou, China. The Laboratory’s education arm also includes an academic publishing house, a graduate school and programs for middle and high school students and teachers. For more information, visit www.cshl.edu
MEDIA CONTACT
Beth Hinshaw Hall, 616.234.5519, [email protected]
