Cryo-EM reveals how ‘911’ molecule helps fix damaged DNA
March 20, 2022

GRAND RAPIDS, Mich. (March 21, 2022) — When something goes wrong during DNA replication, cells call their own version of 911 to pause the process and fix the problem — a failsafe that is critical to maintaining health and staving off disease.
Now, scientists at Van Andel Institute and The Rockefeller University have for the first time revealed how a key piece of this repair process — appropriately called the 911 DNA checkpoint clamp — is recruited to the site of DNA damage. The findings, published today in Nature Structural and Molecular Biology, illuminate new  insights into the way cells ensure genetic instructions are properly passed from one generation of cells to the next. The project was led by the study’s co-corresponding authors Huilin Li, Ph.D., of VAI, and Michael E. O’Donnell, Ph.D., of The Rockefeller University and Howard Hughes Medical Institute.
insights into the way cells ensure genetic instructions are properly passed from one generation of cells to the next. The project was led by the study’s co-corresponding authors Huilin Li, Ph.D., of VAI, and Michael E. O’Donnell, Ph.D., of The Rockefeller University and Howard Hughes Medical Institute.
“DNA damage can have severe consequences, including cancer and other diseases. Because of this, our cells have a host of checks and balances to ensure DNA integrity,” Li said. “Our high-resolution structure of the 911 DNA checkpoint clamp as it interacts with the molecule that loads it onto the DNA strand gives us a detailed look at the essential process of DNA repair. We hope these insights can be leveraged toward the development of new therapeutic strategies for diseases linked to DNA damage.”
Each day, billions of cells in the human body are replaced through cell division, a process by which one cell splits into two. This fundamental function drives growth and facilitates maintenance of tissues such as skin and muscle. A central part of this system is DNA replication, in which our genetic instruction manual is carefully replicated to ensure each cell has an accurate copy.
DNA damage can result from mistakes in this process or through other factors that directly harm DNA, such as exposure to UV light from the sun or carcinogens such as tobacco smoke. When damage occurs, cells have emergency response systems to either stop replication until the problem can be repaired or to kill the cell, thus preventing the incorrect information from being passed on.
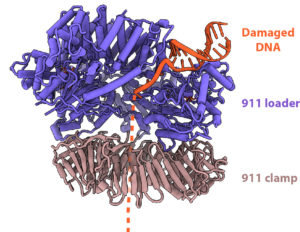
This is where the 911 DNA checkpoint clamp comes in. When DNA damage is detected, the ring-shaped clamp is loaded on the DNA and transported to the site of the error. Once there, it sends a signal to halt cell division while also flagging other repair molecules to remove the damaged DNA and replace it with a corrected sequence.
The structure was determined through use of VAI’s cryo-electron microscopes (cryo-EM), which allow scientists to visualize molecular structures at the atomic level. In the case of the 911 DNA checkpoint clamp, cryo-EM also revealed a surprise: rather than loading onto DNA from the 3’ (or “three prime”) end like all other known DNA clamps, the 911 clamp is loaded onto DNA from the opposite end, called the 5’ (“five prime”) end. This novel and unexpected finding reshapes what we know about DNA replication and sets the stage for further studies in this area.
Other study authors are Fengwei Zheng, Ph.D., of VAI; and Roxana E. Georgescu, Ph.D., and Nina Y. Yao, Ph.D., of The Rockefeller University. Cryo-EM data were collected in collaboration with VAI’s Cryo-EM Core and the David Van Andel Advanced Cryo-Electron Microscopy Suite.
Research reported in this publication was supported by Van Andel Institute (Li), The Rockefeller University (O’Donnell) and the National Institute of General Medical Sciences of the National Institutes of Health under award nos. R01GM115809 (O’Donnell) and R35GM131754 (Li); the Breast Cancer Research Foundation under award no. 20-068 (O’Donnell); and Howard Hughes Medical Institute (O’Donnell). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or other granting organizations.
