Comprehensive study of esophageal cancer reveals several molecular subtypes, provides new insight into increasingly prevalent disease
January 3, 2017

The Cancer Genome Atlas Research Network recommends clinical trials organizers and drug manufacturers focus on newly discovered molecular subtypes
GRAND RAPIDS, Mich. (Jan. 4, 2017) – A comprehensive analysis of 559 esophageal and gastric cancer samples, collected from patients around the world, suggests the two main types of esophageal cancer differ markedly in their molecular characteristics and should be considered separate diseases.
The study, published today in Nature from The Cancer Genome Atlas (TCGA) Research Network, includes two key takeaways. First, upper esophageal cancers more closely resemble cancers of the head and neck, while tumors further down in the esophagus are virtually indistinguishable from a subtype of stomach cancer. Second, cancer clinical trials should group patients according to molecular subtype—in general, grouping lower esophageal tumors with stomach cancers, while evaluating upper esophageal cancers separately.
“These findings add several layers of depth and sophistication to our current understanding of esophageal cancer genomics,” said Adam Bass, M.D., co-leader of TCGA’s esophageal cancer study and physician-scientist at Dana-Farber Cancer Institute. “Our hope is this work settles several long-standing uncertainties in the esophageal cancer field and will serve as the definitive reference manual for researchers and drug developers seeking more effective clinical trials and new treatment approaches.”
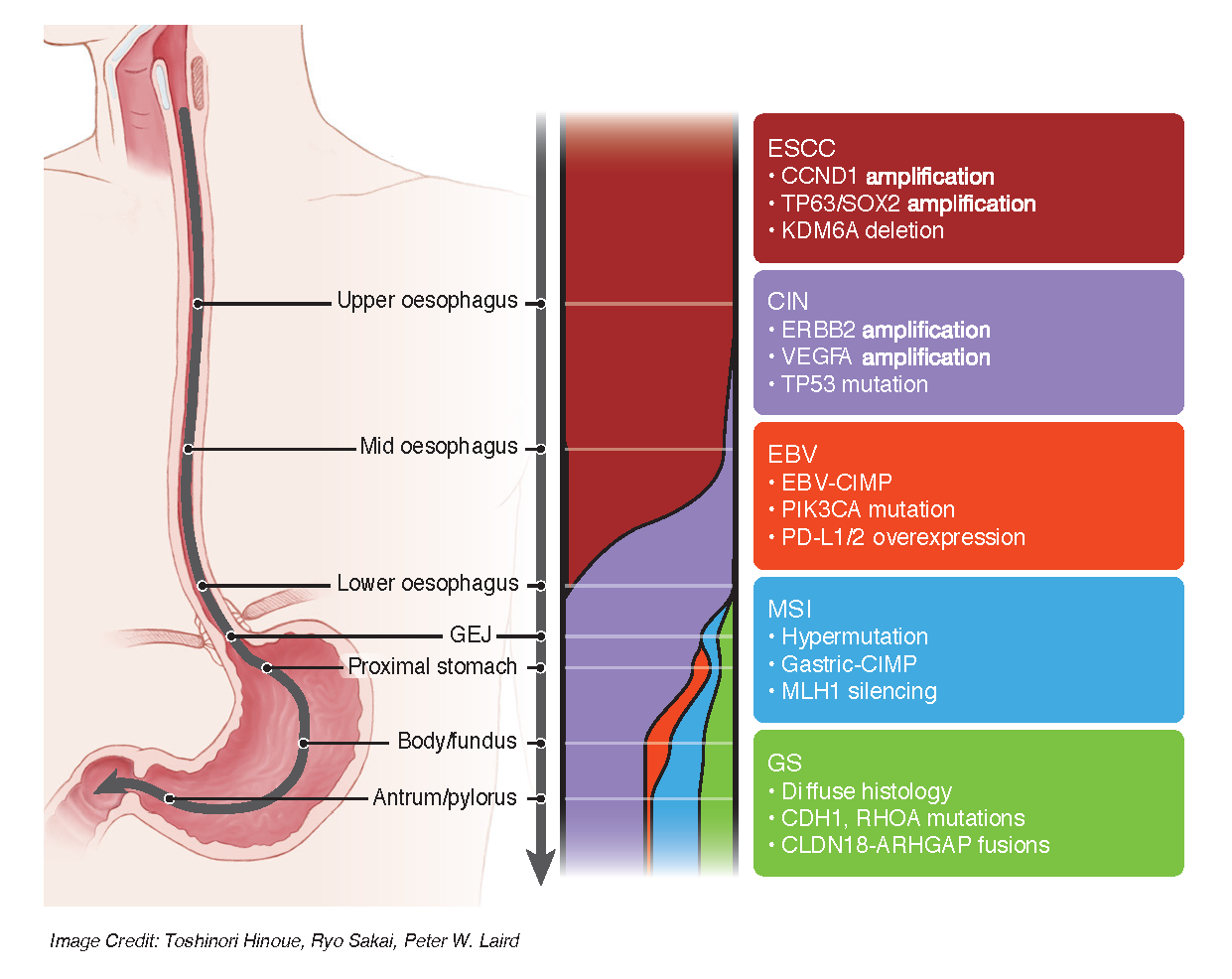 Physicians have known for decades that esophageal cancers, when looked at under the microscope, fall into one of two categories—adenocarcinomas, which resemble stomach or colorectal cancers, and squamous cell carcinomas, which are similar to some lung, skin, and head and neck cancers. What remained unknown was the extent to which adenocarcinomas and squamous esophageal cancers differ molecularly and the relationship between esophageal adenocarcinoma and stomach adenocarcinoma.
Physicians have known for decades that esophageal cancers, when looked at under the microscope, fall into one of two categories—adenocarcinomas, which resemble stomach or colorectal cancers, and squamous cell carcinomas, which are similar to some lung, skin, and head and neck cancers. What remained unknown was the extent to which adenocarcinomas and squamous esophageal cancers differ molecularly and the relationship between esophageal adenocarcinoma and stomach adenocarcinoma.
“We have shown that these clinical subtypes differ profoundly at the molecular level,” said Peter W. Laird, Ph.D., a principal investigator in the international TCGA Research Network and a professor at Van Andel Research Institute. “These findings suggest that whether the tumor originates in the esophagus or the stomach is less relevant than the molecular characteristics of the individual tumors.”
Esophageal cancer represents just 1 percent of new cancer diagnoses in the U.S. However, it kills 4-in-5 patients within five years of diagnosis, and current treatment approaches often fail to help. Additionally, cases of esophageal adenocarcinoma have skyrocketed over the last four decades, increasing seven-fold since the mid-1970s. Within the field, there has been great uncertainty regarding the relationship between this growing burden of esophageal adenocarcinoma and adenocarcinomas that occur in the stomach.
Results from this new report argue against the need to continue to debate the demarcations of esophageal and gastric adenocarcinoma and instead view gastroesophageal adenocarcinoma as a more singular entity, analogous to colorectal cancer. Specifically, this study revealed that esophageal adenocarcinomas have striking molecular similarity to a class of stomach cancers called chromosomally unstable tumors, the hallmark of which are significant structural chromosomal aberrations.
Oncologists say this nuanced view of the disease, including the detailed molecular taxonomy of esophageal adenocarcinomas, will likely change their approach to studies and treatment.
“It is clear from the TCGA data that esophageal squamous and esophageal adenocarcinomas are completely different diseases and should never be included in the same therapeutic trial,” said Yelena Y. Janjigian, M.D., a gastrointestinal oncologist from Memorial Sloan Kettering Cancer Center who contributed samples to the study and served as a co-author. “In esophageal adenocarcinoma, it is likely a combination of pathways and therapeutic strategies that will be successful. The therapeutic significance of these alterations will be explored in follow-up studies.”
Members of the TCGA Research Network team say these studies represent the work of dedicated collaborators, who seek to maximize results in search of new ways to battle cancer.
“Studies from TCGA transcend the work of any one institution or individual,” said Ilya Shmulevich, Ph.D., a principal investigator in the international TCGA Research Network and a professor at the Institute for Systems Biology. “These are massive undertakings that are possible only through contributions from hundreds of specialists and scientists around the world—people dedicate years of their lives to these projects in the hope of finding new treatments for people who are very sick.”
###
About Dana-Farber Cancer Institute
From achieving the first remissions in childhood cancer with chemotherapy in 1948, to developing the very latest new therapies, Dana-Farber Cancer Institute is one of the world’s leading centers of cancer research and treatment. It is the only center ranked in the top 4 of U.S. News and World Report’s Best Hospitals for both adult and pediatric cancer care.
Dana-Farber sits at the center of a wide range of collaborative efforts to reduce the burden of cancer through scientific inquiry, clinical care, education, community engagement, and advocacy. Dana-Farber/Brigham and Women’s Cancer Center provides the latest in cancer care for adults; Dana-Farber/Boston Children’s Cancer and Blood Disorders Center for children. The Dana-Farber/Harvard Cancer Center unites the cancer research efforts of five Harvard academic medical centers and two graduate schools, while Dana-Farber Community Cancer Care provides high quality cancer treatment in communities outside Boston’s Longwood Medical Area.
Dana-Farber is dedicated to a unique, 50/50 balance between cancer research and care, and much of the Institute’s work is dedicated to translating the results of its discovery into new treatments for patients locally and around the world
About Van Andel Institute
Van Andel Institute (VAI) is an independent nonprofit biomedical research and science education organization committed to improving the health and enhancing the lives of current and future generations. Established by Jay and Betty Van Andel in 1996 in Grand Rapids, Michigan, VAI has grown into a premier research and educational institution that supports the work of more than 360 scientists, educators and staff. Van Andel Research Institute (VARI), VAI’s research division, is dedicated to determining the epigenetic, genetic, molecular and cellular origins of cancer, Parkinson’s and other diseases and translating those findings into effective therapies. The Institute’s scientists work in onsite laboratories and participate in collaborative partnerships that span the globe.
About Institute for Systems Biology
The Institute for Systems Biology is a nonprofit biomedical research organization based in Seattle, Washington. It was founded in 2000 by systems biologist Leroy Hood, immunologist Alan Aderem, and protein chemist Ruedi Aebersold. ISB was established on the belief that the conventional models for exploring and funding breakthrough science have not caught up with the real potential of what is possible today. ISB serves as the ultimate environment where scientific collaboration stretches across disciplines and across academic and industrial organizations, where our researchers have the intellectual freedom to challenge the status quo, and where grand visions for breakthroughs in human health inspire a collective drive to achieve the seemingly impossible. Our core values ensure that we always keep our focus on the big ideas that eventually will have the largest impact on human health. ISB is an affiliate of Providence Health & Services.
About the Cancer Genome Research Atlas Network
The Cancer Genome Atlas Research Network represents a collaborative effort of the National Cancer Institute (NCI), National Human Genome Research Institute (NHGRI), and more than 100 medical and research centers on six continents. The Atlas is generating comprehensive, multi-dimensional maps of the key genomic changes in 33 types of cancer that pose immediate public health issues or are poorly understood.

Peter W. Laird, Ph.D.
Professor, Center for Epigenetics
Read the Nature article “Cancer genomics: Spot the difference” by clicking here.
