Clinical trial investigates possible triple threat for lung cancer
November 29, 2017

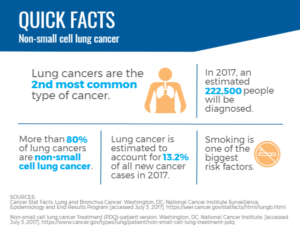
A first-of-its-kind combination of medications may enable the immune system to better combat lung cancer, which claims more lives than any other cancer type and is responsible for more than 220,000 diagnoses this year in the U.S. alone.
“Our immune systems are powerful but sometimes they need help, particularly when faced with diseases like cancer that are specially equipped to evade immune detection,” says Dr. Stephen Baylin, co-leader of the Van Andel Research Institute–Stand Up To Cancer Epigenetics Dream Team, co-head of Cancer Biology at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins and senior author on a paper describing the approach published today in Cell. “Our findings lay the groundwork for an innovative new way to help the body recognize and subsequently destroy cancer cells.”
Cancer can effectively hide from the body’s natural defenses by commandeering so-called epigenetic signals, which help coordinate the immune system’s ability to attack threats and to stand down once the problem is resolved. By hijacking the “off” signal, cancers essentially send an all-clear, preventing the body from going after malignant cells.
Baylin and his collaborators hope to reactivate the “on” signal using epigenetic medications, effectively priming cancer cells to be more susceptible to immunotherapy, which help the immune system fight and kill cancer.
On their own, immunotherapies are already a critical tool in an oncologist’s arsenal. About 20 percent of people with non-small cell lung cancer (the most common type of lung cancer) who are treated with immunotherapies see improvement in their condition, which often is longer lasting than treatment with traditional chemotherapy. The goal, Baylin says, is to grow that number.
“Immunotherapy has revolutionized our ability to combat cancer,” he said. “Combination therapies are the next step in our efforts to continually improve cancer treatment and to give patients more time with a better quality of life.”
The findings described in today’s paper are already being put to the test; they form the basis of a recently launched VARI–SU2C Epigenetics Dream Team clinical trial currently underway at Memorial Sloan Kettering Cancer Center in New York, with additional sites at Johns Hopkins in Baltimore and Fox Chase Cancer Center in Philadelphia slated to open in the coming months.
The trial combines two epigenetic drugs, guadecitabine (Astex Pharmaceuticals) and mocetinostat (Mirati Therapeutics) with the immunotherapy drug pembrolizumab (Keytruda; Merck)—the first time these medications have been used in conjunction with one another. It is supported by a $2.5 million SU2C Catalyst® award and is led by Baylin and Matthew Hellmann, M.D., an oncologist at Memorial Sloan Kettering and also an author on the Cell paper.
Read more about the findings from Johns Hopkins University here and about the VARI–SU2C Epigenetics Dream Team here.
Drs. Baylin and Hellmann are the principal investigators for the SU2C Catalyst award, with Dr. Hellmann serving as the clinical lead for the trial. Additional trial sites include Fox Chase Cancer Center (site lead: Hossein Borghaei, D.O., M.S., chief of the Division of Thoracic Oncology); and the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins and its Bloomberg~Kimmel Institute for Cancer Immunotherapy (site lead: Jarushka Naidoo, M.B.B.Ch., assistant professor of oncology). Hui Shen, Ph.D., VARI assistant professor, and E. John Wherry, Ph.D., director of the Institute for Immunology at University of Pennsylvania will serve as co-investigators for correlative studies.
TRIAL INFORMATION
Trial name: Phase I/Ib study of combined pembrolizumab plus guadecitabine and mocetinostat for patients with advanced NSCLC
ClinicalTrials.gov identifier: NCT03220477
Estimated enrollment: 57 patients
Sites: Memorial Sloan Kettering Cancer Center in New York, New York (enrolling)
Fox Chase Cancer Center in Philadelphia, Pennsylvania
The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins and Bloomberg~Kimmel Institute for Cancer Immunotherapy, Baltimore, Maryland
